Our studies stretch across all levels, from basic research to an in vitro level, from clinical trials with patients to in vivo studies on animal models.
< ResearchWe have different models to study cell interaction both with different types of biomaterials and treatments. This way, we have developed the methodology to study cell migration, proliferation and differentiation, the angiogenesis and biosynthesis of the extracellular matrix, as well as the autocrine and paracrine secretion of growth factors.
Equally, we have developed models that allow us to study the role played by the composition of plasma rich in growth factors in the modulation of the inflammatory stage and the bactericidal properties. In addition to this, plasma rich in growth factors is an interesting alternative to xenogenic products for the ex vivo expansion of stem cells in cell therapy and tissue engineering. Finally, numerous studies are also carried out in an aim to optimise the results of this personalised therapy, including the evaluation of possible interferences due to the consumption of regular medicines.
For BTI, our patients and their well-being are always the focus of our research process. For this reason, our focus has a clearly biological component that comes from profound knowledge of the tissue regeneration processes and is based on optimising and adapting the treatment to the patient's clinical situation.
In this regard, we carry out several lines of research to develop/evaluate surgical techniques and/or autologous treatments in tissue regeneration. Preservation of the post-extraction socket is a good model for evaluating the capacity of different clinical alternatives (both in materials and protocols) when accelerating bone regeneration, curing soft tissue and reducing patients' discomfort.
We also study both the prevention and treatment of the occurrence of complications associated with extraction, such as dry alveolitis or osteonecrosis of the maxilla associated with medicines (MRONJ). Therefore, the use of autologous growth factors allows us to advance in different lines of research in the field of periodontal regeneration, peri-apical surgery, regenerative endodontics, pre-implant surgery for bone augmentation and advanced bone regeneration therapy. Also, the use of autologous proteins from blood in the treatment of erosive oral lichen planus and other ulcerative conditions opens a new horizon in clinical and translational research.
Temporomandibular joint disorders (ATM) are often associated with chronic pain and imply a handicap for everyday activities, reducing patients' quality of life. In this context, we are carrying out several lines of research to design new treatments revolving around the intra-articular injection of PRGF.
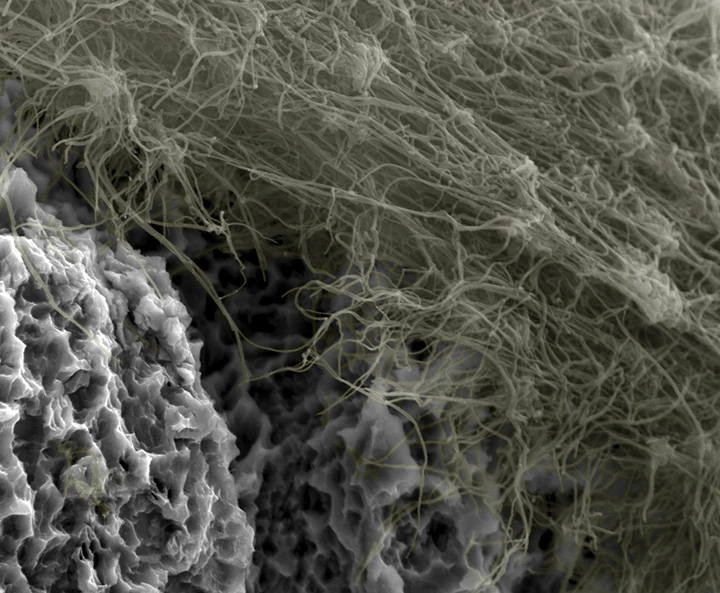
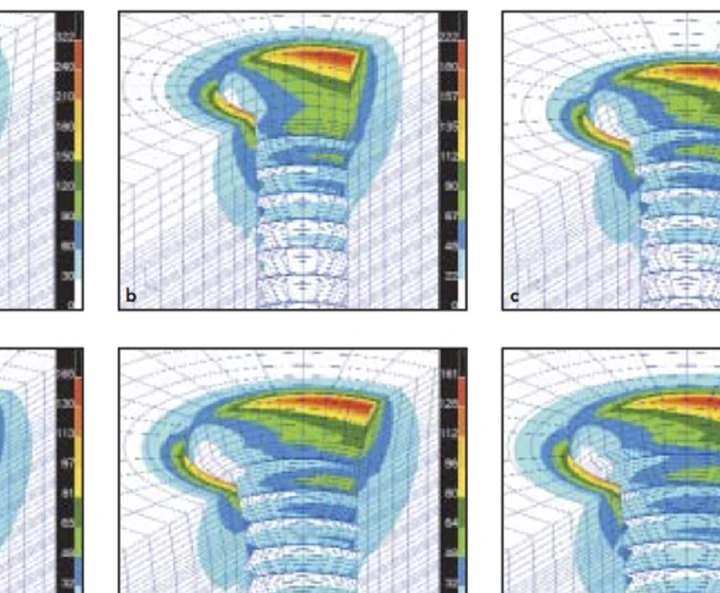
As international role models in the field of implantology, we are constantly evaluating the predictability of the BTI implant system. We are especially interested in developing minimally invasive predictive protocols that have a clear biological focus. Therefore, the study of risk factors for the failure of implant treatments provides us with ideas for studying biomechanics and the surface of dental implants. This means that the designs of BTI dental implants, at the macro and micro level, are optimised.
We carry out research in connection with the "Bio-Block" concept that brings together biomechanics, biology and clinical elements. We therefore encourage and participate in lines of research that enable the optimisation of the prosthetic design. Furthermore, the digitisation of oral implantology is an extremely promising line for facilitating clinical work, improving its results and preparing for facing new epidemiological challenges.
There are currently many ocular disorders that are characterised by limitations in the repair of affected tissues and which present themselves as unresolved pathologies or post-operative complications. At BTI, we're searching for a solution to these problems through different research projects. Likewise, over the last decade, our research team has been working on the development of autologous eye drops for the ocular surface that have a standardised, predictable, personalised and stable protocol for up to 1 year.
This line of research involves three main areas: ocular surface diseases, ophthalmological surgery and ocular fundus pathologies.
The range of pathologies that can affect the ocular surface is very varied and has different levels of severity, from mild to very serious. Among them, we find: dry eye syndrome (DES), persistent epithelial defects (PEDs), disorders of the ocular surface and scarring conjunctivitis. We are carrying out different studies on all of them. We are also investigating the use of fibrin membrane as a three-dimensional mesh for treating surgical wounds and adjuvant treatment following glaucoma surgery, persistent corneal ulcer, perforation of the cornea, surface keratoplasty and pterygium excision. Finally, it's worth highlighting that, nowadays, several ocular fundus pathologies are orphan when it comes to treatment and we are carrying out research to offer autologous alternatives taken from the patient's blood.
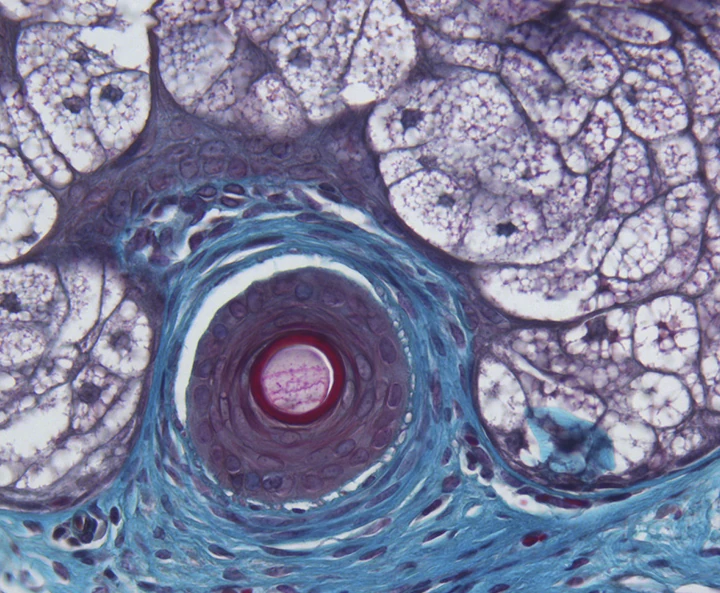
Over the last few decades, the progressive increase of the average age of the population has brought with it an increase in the prevalence of chronic and degenerative skin problems. In this sense, the field of regenerative dermatology has become a key discipline in the development of innovative technologies in biomedical material. The development of autologous therapies based on products taken from the patient's blood has had a resounding effect on the area of skin regeneration due, to a large extent, to its therapeutic potential and its profile in terms of biosafety.
Wounds that are difficult to cure are often associated with underlying and systemic pathologies and can lead to an ulcer becoming chronic. These wounds suffer from an unbalance in cellular and protein components that provoke poor regulation in the closure of the wound. Therapy with autologous proteins is a research field that seeks to accelerate the formation of healthy granulation tissue that is liked to a greater epithelialisation ratio and, therefore, reduces the hospitalisation period and the economic cost associated with the treatment.
Progressive ageing and deterioration of the skin is a multifactorial process due to intrinsic (age, eating habits) and extrinsic (exposure to ultraviolet light, pollution, toxic agents) causes. Characterised by the gradual decrease in the functional capacity of the tissue, causing physiological alterations that affect both cellular and structural components of the skin. In this regard, our research is developed in in vitro and ex vitro models. In this sense, using autologous proteins, we're seeking to counteract ageing of the skin promoting the synthesis of key components in the extracellular matrix, such as collagen or hyaluronic acid. Also, cell activity and viability are significantly increased which, in turn, improves hydration and the quality of the surrounding tissue.
Self-esteem and image play a key role in our social perception, personal interaction and psychosocial functioning. Gradual hair loss can lead to serious emotional disorders and significantly reduce patients' quality of life. Although some molecular mechanisms involved in capillary degeneration are known, the specific pathophysiological causes of alopecia remain unclear. In this sense, with this line of research, using autologous proteins, we seek not only to reactivate bulbar activity but also to improve the quality of the surrounding skin tissue, which is what nourishes and provides biomechanical support for the hair follicle.
Our research group was a world pioneer in beginning to use blood-based autologous therapies in traumatology and sports medicine. Nowadays, we continue to develop several research projects with which we aim to minimise the impact of injuries and illnesses of the locomotive system in physical activity with a double objective: to accelerate functional recovery and reduce the patient's level of suffering and frustration.
We approach our research into articular pathologies comprehensively, considering all elements of articulation. Our objective is to reduce pain and inflammation and improve the patient's mobility.
For chondral injuries, our line of research seeks to improve the repair of cartilage and decrease post-operative inflammatory signs after surgical interventions for the treatment of osteochondritis dissecans, chondral injuries with a non-valuable fragment, extensive chondral injuries and in the treatment of avascular necrosis of the hip.
In this line of research, we seek an autologous therapy characterised by its outpatient application which reduces the recovery time after this type of injuries and may be an alternative to reconstructive surgery. Some examples are chronic tendinopathy, acute tendon rupture, chronic tendon rupture in patients with a lower functional demand and ligament rupture, such as the anterior cruciate ligament.
Muscular injuries make up 35-45% of sports injuries. For an athlete, these injuries represent a significant loss in terms of training sessions and competitions. There are currently no pharmaceuticals available to accelerate the recovery of muscle function after an injury. Therefore, in this line of research, we're investigating autologous treatment with growth factors to reduce the recovery time after these types of injuries as well as the rate of recurrences.
The failure of a fracture to unite happens when there's a fault in the normal biological bone healing process. Our objective in this line of research is to develop therapeutic strategies with a dual effect: a preventative effect to minimise the prevalence of this complication and a therapeutic effect that overcomes the failure in the bone regenerative process when it occurs.
In this research line, we're investigating the combination of minimally invasive techniques and autologous growth factors in herniated discs, protrusions, degenerative disk injuries or to treat arthritis of the cervical and lumbar spine.

